Ricochet Science Macromolecules Series
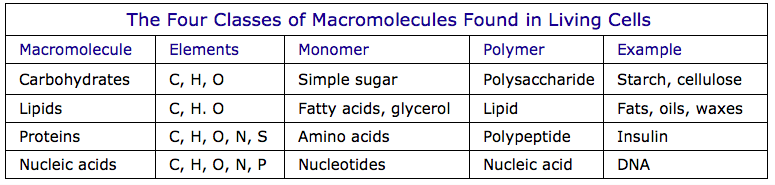
This series of videos covers carbohydrates and lipids in somewhat more depth than proteins and nucleic acids. Proteins and especially nucleic acids will be featured in more detail in speparate DNA Basics pages. All of the RicochetScience material is presented very clearly. This series will be invaluable for students studying for exams. As a bonus, the series provides a good framework for understanding the terminology of dietary guidelines and recommendations.
Together they total about 20 minutes.
Biomolecules & functional GroupsRicochetScience (3 Nov 2015) Biomolecules and Functional Groups [Video file] retrieved from https://youtu.be/OGD3q1eQ1TE |
|
CarbohydratesRicochetScience (3 Nov 2015) Carbohydrates [Video file] retrieved from https://youtu.be/LeOUIXbFyqk |
|
LipidsRicochetScience (12 Apr 2016) Lipids [Video file] retrieved from https://youtu.be/5BBYBRWzsLA |
|
ProteinsRicochetScience (13 Nov 2015) Proteins [Video file] Retrieved from https://youtu.be/AUMJwjLXh1M |
|
Nucleic AcidsRicochetScience (1 Oct 2015) Nucleic Acids [Video file] retrieved from https://youtu.be/MA-ouz1LtpM |